The molecular mechanisms underlying long-term memory impairment in early- and late-onset mouse models of ADRD.
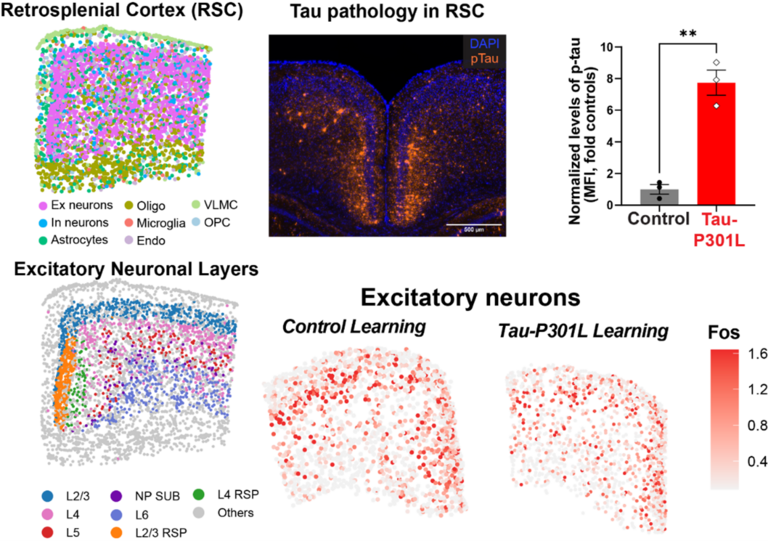
Deficits in cognitive function and memory represent a debilitating aspect of neurodegenerative diseases, leading to long-term disability, enormous suffering for individuals and their families, and substantial socioeconomic costs. The pathological hallmarks of Alzheimer's disease and related dementias (ADRDs) include tau tangles and β-amyloid (Aβ) plaques, with tau tangles observed in more than 55 million individuals worldwide. Mutant tau mouse models of ADRD exhibit impaired long-term memory. Long-term memory consolidation requires the induction of gene expression in a specific temporal pattern. Dysfunction in gene expression can lead to memory impairment and may be dysregulated in disease conditions, although the precise mechanisms by which this occurs are unclear. We use multifaceted approaches, including single-nuclei RNA sequencing, chromatin accessibility analysis, spatial transcriptomics, spatial epigenetics, gene manipulation, and behavioral pharmacology, to study how molecular mechanisms of memory function in two brain regions—the dorsal hippocampus and the retrosplenial cortex—and their relation to ADRD-related memory loss.
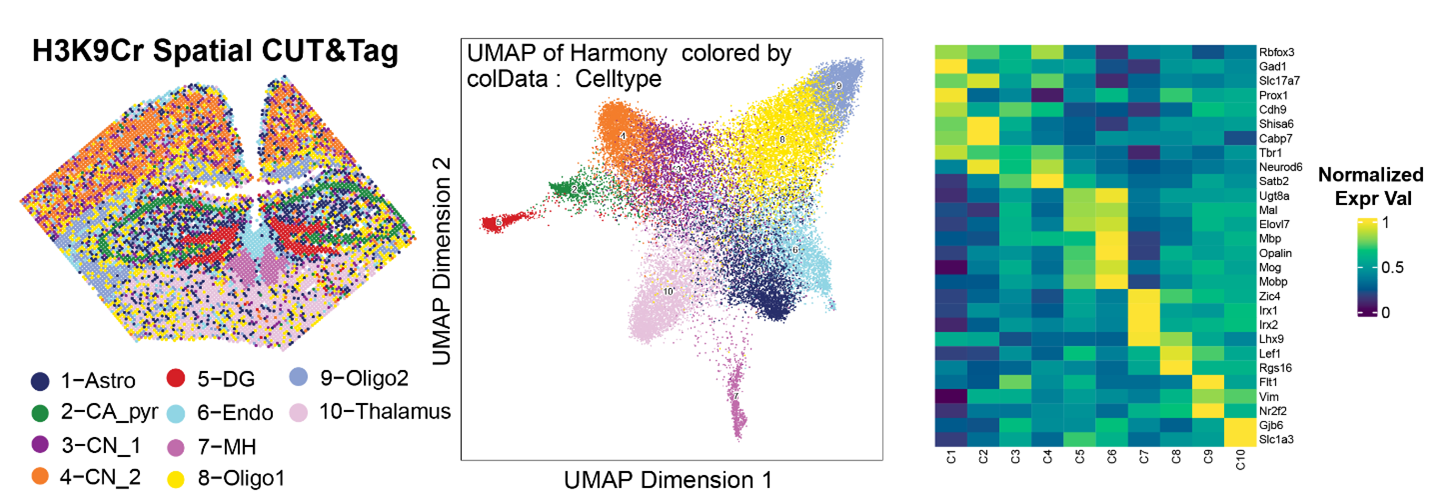
Role of histone crotonylation modifications in memory and neurodegeneration.
Memory consolidation depends on epigenetic modifications to DNA and histones that underlie gene transcription. Recent studies have revealed that a subset of histone residues is susceptible to non-acetyl short-chain lysine acylation that is mechanistically and functionally distinct from histone acetylation. We found that blocking histone lysine crotonylation (Kcr) modification impairs memory, while enhancing this epigenetic modification enhances memory and gene expression. Using Spatial CUT&Tag, Spatial ATAC-Seq, combined with Xenium spatial transcriptomic profiling at single-cell resolution, we are currently investigating the roles of specific histone lysine crotonylation modifications in memory and neurodegeneration. We are investigating the relationship between these histone post-translational modifications and neuropathology in Alzheimer's disease and related disorders (ADRD).
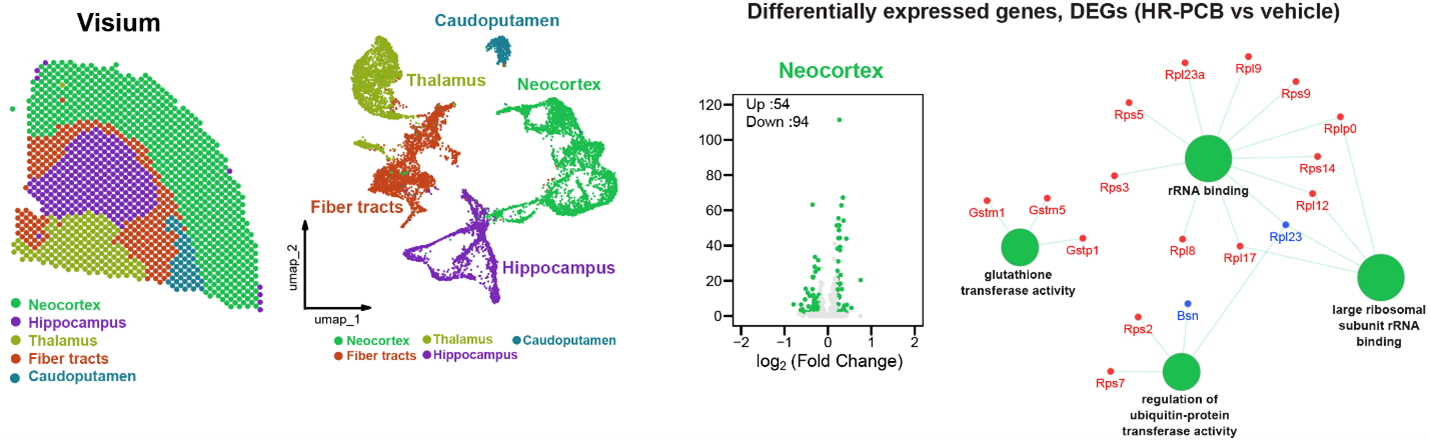
Investigate how exposure to environmental toxins adversely affects ADRD.
We use mouse models of ADRD to examine how environmental factors influence the development and progression of ADRD risk. We show that a human-relevant polychlorinated biphenyls (PCB) impairs long-term spatial memory in adult mice. Spatial transcriptomic analysis revealed gene-expression signatures influenced by PCB in susceptible brain regions of mice. Ongoing research is assessing how human-relevant PCB mixtures alter the development and progression of ADRD-like phenotypes in mouse models of ADRD. Identifying PCBs as environmental risk factors that alter ADRD-related outcomes will lay the groundwork for mechanistic studies and inform translational studies to treat ADRD impacted by environmental toxicants within a precision environmental health framework.